Decoding a Low‑Cost Microfluidic Brain‑on‑a‑Chip Platform for Neuronal Culture and Drug Screening
Category: Neuroscience | Bioengineering |
Original Research Paper:
Crafting Precision: Design and Fabrication of a Xurography‑Driven Microfluidic Platform for Exploring Neuron Culture and Targeted Drug Screening
Authors: Subhadra Nandi, Arijit Bera, Satyajit Ghosh, Anindyasundar Adak, Rajsekhar Roy, and Surajit Ghosh
Journal: ACS Chemical Neuroscience (2025)
DOI: 10.1021/acschemneuro.5c00016
ACS Chemical Neuroscience (2025)
Paper Decoded — Key Takeaway Up Front
This study shows that advanced neuronal microfluidic platforms do not necessarily require cleanrooms, expensive photolithography, or elite infrastructure. By using xurography (razor‑writing)—a low‑cost cutting‑plotter‑based fabrication method—the authors built a reproducible microfluidic device that supports 3D neuronal culture, astrocyte–neuron coculture, and proof‑of‑concept Alzheimer’s drug screening.
Importantly, this work does not claim a cure for Alzheimer’s disease. Instead, it introduces an accessible experimental platform that can accelerate early‑stage neurotherapeutic research, especially in resource‑limited laboratories.
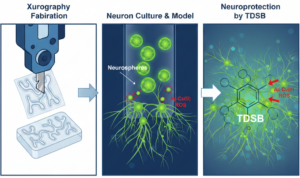
The Core Problem: Why Brain Research Is So Hard (and Expensive)
The human brain is a highly organized, three‑dimensional network of interacting cells exposed to tightly regulated chemical and mechanical cues. Yet, much of neurobiological research still relies on two‑dimensional cell cultures grown on flat plastic dishes. These simplified models fail to capture realistic cell–cell interactions, fluid dynamics, and spatial organization, often leading to misleading biological responses.
To overcome this, researchers increasingly use microfluidic or “lab‑on‑a‑chip” systems, which allow precise control over microenvironments and multicellular organization. However, most microfluidic devices are fabricated using soft lithography, a technique that requires:
- Cleanroom facilities
- Photomasks and UV lithography systems
- Specialized technical expertise
- High setup and maintenance costs
These requirements place advanced neuronal microfluidics out of reach for many academic labs, particularly in developing countries.
The Central Innovation: Xurography‑Based Microfluidic Fabrication
What Is Xurography?
Xurography—often called razor writing—is a rapid prototyping technique in which a computer‑controlled cutting plotter precisely cuts microchannel patterns into thin adhesive films. These patterned films serve as molds for casting PDMS (polydimethylsiloxane), a transparent and biocompatible silicone commonly used in microfluidics.
Unlike photolithography, Xurography:
- Requires no cleanroom
- Uses inexpensive, desktop‑scale equipment
- Allows rapid design iteration
In this study, the authors demonstrate that Xurography can achieve ~150 μm channel resolution, which—while lower than photolithographic methods—is sufficient for many neuronal culture applications.
A Critical Design Insight: Why the Right Tape Matters
The success of low‑cost fabrication hinged on an often‑overlooked detail: the choice of mold material.
The authors compared multiple adhesive films:
- PVC tape and Scotch tape failed at elevated curing temperatures, releasing residues and producing uneven PDMS surfaces that compromised imaging and cell growth.
- Kapton tape, in contrast, proved thermally stable, chemically inert, and optically suitable.
Kapton‑based molds produced smooth, transparent microchannels and could be reused after simple ethanol cleaning, significantly reducing cost and material waste. This optimization highlights how small engineering choices can have outsized biological consequences.
Device Geometry: When Engineering Dictates Biology
Three microfluidic geometries were fabricated and evaluated:
- H‑shaped
- X‑shaped
- C‑shaped (straight channel)
Using nanoparticle tracer experiments, the authors quantitatively analyzed fluid flow behavior. The results were decisive:
- H‑ and X‑shaped devices generated higher flow velocities, leading to cell washout and channel clogging.
- The C‑shaped design provided stable, low‑shear conditions ideal for static neuronal culture.
Key lesson: In microfluidic systems, device geometry is not aesthetic—it directly determines biological outcome.
From Flat Cultures to 3D Brain‑Relevant Models
Primary Neurons and Neurospheres
When primary rat hippocampal neurons were seeded at high density, they self‑assembled into three‑dimensional neurospheres within 48 hours. These structures better resemble in vivo neural organization than monolayer cultures and remained viable for up to 10 days in vitro.
At lower densities, neurons formed healthy monolayers with preserved morphology, demonstrating the platform’s versatility.
Astrocyte–Neuron Coculture
To model multicellular brain interactions, the authors developed a vertically stratified coculture system:
- Astrocytes were cultured on the upper PDMS surface
- Neurons were cultured on the lower glass surface
- The two populations were separated by ~100 μm
This configuration allowed biochemical communication while maintaining spatial separation, enabling controlled studies of neuron–glia interactions—an essential but often neglected aspect of neurodegenerative disease research.
Proof of Concept: Alzheimer’s Drug Screening on the Chip
To demonstrate practical utility, the platform was used to test a rationally designed compound: TDSB (L‑tryptophan‑1‑hydroxy‑2‑naphthaldehyde‑dopamine Schiff base).
Why TDSB?
Alzheimer’s disease is multifactorial, involving:
- Amyloid‑β aggregation
- Metal‑induced oxidative stress
- Mitochondrial dysfunction
Rather than targeting a single pathway, TDSB was designed as a multi‑target molecule.
Key Findings
- Amyloid‑β inhibition: ~82% inhibition of fibril formation at 40 μM; ~75% disaggregation of pre‑formed fibrils at 20 μM
- Metal chelation: Selective binding to Cu(II), disrupting redox cycling
- Oxidative stress reduction: >95% suppression of hydroxyl radical generation
- Cellular protection: Restoration of neuronal viability and mitochondrial membrane potential
In the astrocyte–neuron coculture model, TDSB not only protected neurons from amyloid toxicity but also suppressed astrocytic activation, preserving a healthier multicellular environment.
Why This Paper Matters
This study delivers two important contributions:
1. Democratizing Neuroengineering
By validating Xurography as a serious fabrication strategy, the work lowers the entry barrier to advanced neuronal microfluidics. Laboratories without cleanrooms can now explore physiologically relevant brain models using affordable tools.
2. Improving Early‑Stage Drug Evaluation
Testing therapeutics in 3D, multicellular systems provides insights that are often missed in conventional assays. This platform offers a realistic and scalable bridge between biochemical screening and animal studies.
Limitations and Responsible Interpretation
The authors appropriately acknowledge limitations:
- Resolution (~150 μm) is lower than photolithography
- Fabrication quality can vary with cutting conditions
- The system is optimized for static culture, not continuous perfusion
- Long‑term scalability and clogging control require further optimization
Importantly, this work represents an enabling platform, not a clinical solution.
Final Decoded Insight
Innovation in biomedical research does not always come from higher precision or higher cost—but from thoughtful engineering choices that balance accessibility with biological rigor.
For researchers in neuroscience, diagnostics, and bioengineering, this paper serves as a practical roadmap for building meaningful experimental systems without prohibitive infrastructure.:
Want more papers decoded like this?
Follow Research Paper Decoded for unbiased, technically accurate explanations of cutting‑edge research—without unnecessary jargon or hype.
Disclaimer:
This article decodes published research for educational purposes and does not provide medical or clinical advice.

Pingback: Xurography Decoded: The Cheapest Way to Build Labs-on-a-Chip for Biology - Research Paper Decoded