Paper Details
Title: Crafting Precision: Design and Fabrication of a Xurography-Driven Microfluidic Platform for Exploring Neuron Culture and Targeted Drug Screening
Authors: Subhadra Nandi, Arijit Bera, Satyajit Ghosh, Anindyasundar Adak, Rajsekhar Roy, and Surajit Ghosh
Journal: ACS Chemical Neuroscience (2025)
DOI: 10.1021/acschemneuro.5c00016
Why This Paper Matters
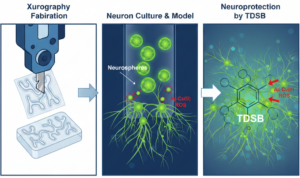
The Nandi et al. paper addresses a critical gap in neurodegenerative disease research: the democratization of microfluidic technology. While microfluidics has revolutionized our understanding of neuronal behavior and enabled sophisticated drug screening, most fabrication methods require expensive equipment, specialized cleanroom facilities, and significant expertise. This study presents Xurography—also known as “razor writing”—as a low-cost, rapid prototyping alternative that maintains scientific rigor while dramatically reducing barriers to entry for resource-limited research institutions.
For researchers in countries like India, or any institution with budget constraints, this work opens unprecedented opportunities to establish cutting-edge neurobiological platforms without breaking the bank.
The Innovation: Xurography for Microfluidic Fabrication
What is Xurography?
Xurography is a cutting-plotter-based fabrication technique where computer-controlled cutting heads precisely carve microchannel patterns from adhesive polymer films. The approach, first introduced in 2005, has seen limited application in neurobiological research despite its potential. This paper represents one of the first comprehensive demonstrations of xurography-based microfluidic devices specifically designed for neural culture and disease modeling.
Why This Matters for Device Design
Traditional microfluidic fabrication methods like soft lithography require:
- Cleanroom facilities
- Expensive photomasks and equipment
- Multiple specialized steps
- High per-unit costs
The authors optimized xurography to achieve:
- 150 μm channel resolutionusing PDMS with controlled cutting force (188-620 milliNewton)
- Reusable Kapton tape moldsthat eliminate material waste
- Desktop fabricationusing a Cricut cutting machine
- Standardized protocolsto ensure reproducibility across batches
A particularly elegant design choice was the selection of Kapton tape as the substrate after comparing PVC and Scotch tape alternatives. At elevated baking temperatures (80°C), PVC and Scotch tape released oily residues that compromised PDMS surface quality and optical clarity. Kapton tape proved thermally stable and biocompatible, enabling both clear imaging and reusability after simple ethanol cleaning—a small but significant optimization that researchers can replicate.
Device Characterization and Optimization
Flow Dynamics and Device Selection
The team fabricated three distinct device geometries: H-shaped, X-shaped, and C-shaped (straight channel) designs. Here’s where experimental rigor shines through.
Using nanoparticle tracer studies, they quantified fluid flow dynamics in each design:
- H-shaped and X-shaped devicesexhibited rapid flow that washed out primary neurons or caused channel clogging
- C-shaped devicedemonstrated optimal flow characteristics for static cell culture (their intended application)
This led to a critical insight: device geometry profoundly influences biological outcomes. The C-shaped design wasn’t chosen because it looked simpler—it was selected because quantitative flow analysis predicted superior cellular adhesion and neurosphere formation, which they subsequently validated experimentally.
Cytocompatibility: The Three-Pronged Validation
The authors demonstrated device suitability through:
- Primary Neuron Culture & Neurosphere Generation
- Primary rat hippocampal neurons cultured at high density formed 3D neurospheres within 2 days
- Neurospheres maintained viability for 10 days in vitro (DIV)
- Low-density seeding produced monolayers with intact neuronal morphology
- β-III Tubulin (TUJ1) immunostaining confirmed neuronal identity
- SH-SY5Y Cell Line Culture and Differentiation
- Cells attached within 4 hours of seeding
- Reached confluency within 2 DIV
- Successfully differentiated using 10 μM retinoic acid + 50 ng/mL BDNF
- Live/Dead assays (Calcein-AM/PI staining) confirmed high viability at both low (10,000 cells) and high (50,000 cells) seeding densities
- Astrocyte-Neuron Coculture
- Vertically stratified coculture with ~100 μm separation
- GFAP+ astrocytes maintained stellate morphology
- TUJ1+ neurons exhibited healthy neurite arborization
- Z-stack imaging confirmed distinct cellular layers
This three-part validation framework is exemplary: each model system builds complexity while maintaining clear interpretation of results.
The Real Test: TDSB—A Multi-Targeted Alzheimer’s Therapeutic
The paper’s most compelling contribution emerges in the second half: proof-of-concept drug screening using a rationally designed compound.
TDSB: Design Philosophy
The authors synthesized TDSB (L-tryptophan-1-hydroxy-2-naphthaldehyde-Dopamine Schiff Base), a three-in-one molecule targeting distinct Alzheimer’s pathology mechanisms:
- Metal Chelation: 1-hydroxy-2-naphthaldehyde moiety
- Amyloid-β Fibril Disruption: Tryptophan residue (evidence from prior studies)
- Antioxidant Activity: Dopamine component
This represents sophisticated medicinal chemistry—not just combining random components, but carefully selecting pharmacophores targeting validated therapeutic targets in Alzheimer’s disease.
Key Biochemical Findings
Amyloid-β Aggregation Inhibition:
- At 20 μM: TDSB disaggregated ~75% of Aβ42 fibrils (IC50 = 3.9 μM)
- At 40 μM: ~82% inhibition
- Outperformed positive control Apigenin (60% at 20 μM)
- Dot blot assay confirmed dose-dependent aggregate reduction
Metal Chelation & ROS Prevention:
- Restored Tyrosine-10 fluorescence in Aβ42-Cu(II) complexes (1:1 binding stoichiometry)
- Displayed remarkable selectivity for Cu(II) over other physiological metals (Zn, Co, Ni)
- Inhibited hydroxyl radical generation in Fenton-type redox cycle by 95%+
Cellular Efficacy:
- SH-SY5Y-derived neurons treated with Aβ42-Cu(II) showed 55% viability
- With 20 μM TDSB: restored to ~80% viability
- DCFDA assays confirmed ROS suppression (***p < 0.001)
- JC-1 flow cytometry demonstrated restored mitochondrial membrane potential
The Microfluidic Validation: Where Biology Meets Platform
This is where the paper transcends a typical drug screening study. Using their xurography-fabricated coculture platform, the authors demonstrated:
In untreated controls: Astrocytes displayed stellate morphology; neurons showed robust arborization
Upon Aβ42 exposure: Neuronal processes retracted, soma condensed; astrocytes activated (morphological sign of gliosis)
With TDSB treatment: Morphological toxicity reversed; astrocyte activation suppressed; neuronal architecture recovered
This microscopy-based validation in a physiologically relevant coculture system is far more persuasive than isolated biochemical assays. It’s the difference between showing a compound works in a test tube versus showing it protects neural networks.
Technical Strengths and Innovation Highlights
- Rigorous Device Optimization: Flow analysis → device selection → biological validation follows gold-standard methodology
- Cost Transparency: The entire fabrication workflow is accessible to any lab with a $200-400 cutting plotter
- Rational Drug Design: TDSB wasn’t arbitrary; each component targets distinct Alzheimer’s mechanisms
- Multi-Scale Validation: From biochemical assays → cell viability → tissue-level coculture
- Reproducibility Focus: Explicit protocols for Kapton tape preparation, cutting parameters, and standardized postfabrication treatment
Limitations and Future Directions
The authors thoughtfully acknowledge constraints:
- Resolution Limits: 150 μm is orders of magnitude larger than standard photolithography; complex micro-mixer designs remain challenging
- Operator-Dependent Variability: Cutting speed, blade sharpness, and material storage affect reproducibility
- Static Culture Model: The C-shaped design optimizes static culture; dynamic flow-based cultures require different geometries
- Scalability: Single-device production differs from manufacturing hundreds of units with consistent quality
- Technical Issues: Sample loss, cross-contamination, and clogging require further protocol development
These honest assessments strengthen rather than weaken the paper—they define the scope of application and suggest future research directions.
Broader Impact and Research Implications
For Diagnostic/Therapeutic Development
This platform could accelerate neurotherapeutic screening in settings where budget constraints have historically limited research capacity. Developing countries, smaller research institutions, and startup biotech firms gain access to sophisticated cell culture systems previously reserved for well-funded laboratories.
For Alzheimer’s Research
The successful integration of TDSB screening in a physiologically relevant coculture model demonstrates proof-of-concept for multi-targeted therapeutic validation. The astrocyte-neuron interaction aspect is particularly valuable—astrocytic dysfunction plays an underappreciated role in Alzheimer’s pathology.
For Microfluidic Standardization
By thoroughly characterizing Xurography’s capabilities and limitations, this work contributes essential data for the emerging “democratized microfluidics” movement. Future studies can build on this foundation rather than starting from scratch.
Critical Questions and Extensions
- How does TDSB perform in vivo? The paper establishes efficacy in coculture models, but animal studies are essential before clinical translation.
- Can this platform model other neurodegenerative diseases?Parkinson’s, Huntington’s—the framework is generalizable.
- What’s the long-term stability of xurography-fabricated devices?Weeks? Months? This affects commercial viability.
- Can microfluidic and xurography approaches integrate with organoid technology?Recent advances in brain organoids suggest exciting hybrid possibilities.
Why This Deserves Your Attention
This paper exemplifies how thoughtful engineering combined with rigorous biology can democratize cutting-edge research. It’s not groundbreaking in the sense of a paradigm-shifting discovery, but it’s enabling—it removes barriers and opens doors.
For biotech researchers in resource-limited settings, this is a roadmap. For pharmaceutical companies seeking cost-effective screening platforms, this is a validation of alternative fabrication strategies. For the global Alzheimer’s research community, TDSB represents a promising lead compound warranting further investigation.
The marriage of accessible fabrication with meaningful drug discovery makes this a paper worth reading deeply and implementing practically.
Key Takeaways for Your Research
- Xurography is viable for serious neuroscience research, not just prototyping
- Device geometry optimization requires quantitative flow analysis, not intuition
- Multi-targeted compounds addressing distinct pathology mechanismsmay be the future of neurotherapeutic design
Astrocyte-neuron crosstalk is therapeutically relevant and practically achievable in microfluidic systems
